"Measure what is measurable, and make measurable what is not so" - Galileo Galilei |
Week 4 Resources/ Assets |
|
Updated September 13, 2018
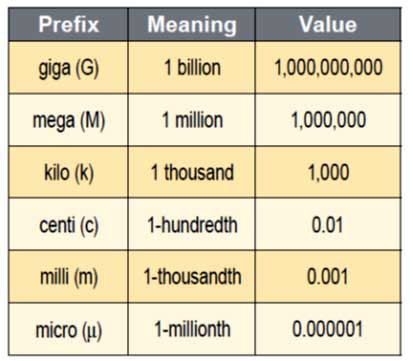
Profound and almost prophetic words spoken by Galileo! We'll address the importance of measurement below, but if you are seeking the landing page for weekly announcements for chemistry class... this is it! Please make sure you READ everything on this page or you will miss important details. If you need to go back to a particular weeks information for any reason, the weekly Announcements pages will appear under the Trimester 1 tab. Administrative details... read this young chemists! :) On the class Discussion Board (right), there is a forum for students to ask general questions about assignments, labs, quizzes, due dates, etc. If you have a question, please feel free to post it, because it just may help other students who have the same question. Also, if you need some further assistance in solving a particular problem, then please upload to your mobile device the app called Appear.in - which permits video chatting - for up to eight students at a time. I have created a room where we can meet to solve problems, called veritas-chemistry. If you are interested in getting a little more help than what can be offered through an email, please let me know by email, and we can arrange a time to meet in the veritas-chemistry room. Looking Ahead to Week 4 Now that you have a good idea as to: (1) what has driven the field of chemistry (development of materials and harnessing of energy), (2) the different types of substances (pure substances - elements and compounds; mixtures - homogeneous and heterogeneous), (3) separation techniques (filtration, paper chromatography, recrystallization), (4) the scientific method, and, (5) conservation of mass laws, we now turn our attention to the issue of measurement. Start Chapter 3 - Scientific Measurement Please take the time this weekend to peruse through Chapter 3 and answer as many questions as you can in the homework (linked right). Please note that I have also linked the Answer Key for Chapter 3. Much of this should be a review from your previous math classes. When we return to class on Monday, we will hit the ground running with the Significant Figures Laboratory (linked right). Significant figures Galileo's quip was truly prophetic of where the sciences (or natural philosophy in his day) would advance. Today, data (singular, datum) by it's very definition entails measurements in one form or another. Qualitative measurements of a few decades ago, have become quantitative measurements today with the advancement of highly-refined tools. In spite of the development of these tools, they still are beset with some degree of inaccuracy. This is where significant digits come in. I will say nothing else here about significant digits - I have uploaded a video at right that addresses their rationale and how to perform operations with them. I will not dedicate much more time to Significant Figures in class - you need to watch this video in order to complete the calculations from the lab you started on Tuesday. Please remind yourselves of the definitions for precision, accuracy, and error (pp. 54, 55 in text). Scientific Notation Hopefully you are now comfortable with manipulating figures expressed in scientific notation. Please make sure you are adept at keying these numbers into your calculators! If you need additional help with scientific notation, then I encourage you to watch the Tyler Dewitt videos at right. For next Tuesday's quiz, you will need to perform calculations with scientific notation. SI units/ Metric system In the olden days, body parts provided convenient units of length. For one thing, they are pretty handy, since you are never out and about without thumbs (hopefully), your feet, noses or arms. Unfortunately, the measurements provided by these measuring devices are not altogether reliable or consistent. Today, we use standardized measures with units derived largely from the metric system. Please do familiarize yourself with SI units and the metric system. When reporting data in the worlds of chemistry and physics, we typically use the units as follows:
Finally, in Chapter 3, you will learn about density, which, like temperature, boiling point, melting point, etc., is a physical property of matter. Density gives to us an idea of how much matter a substance has per unit of volume. Water has a density of 1 gram/ cubic centimeter, whereas aluminum has a density of 2.7 g/ cubic centimeter. Knowing this, do you think a cubic sample of aluminum would sink or float in water? To prepare for classes next week, please make sure you do the following:
|
Scientific Notation videos by Tyler DeWitt (optional) |